|
If 100 cal of heat is added to one g of each metal at 25*C, what is the expected result?Ī) the copper will reach a higher temperatureī) the silver will reach a higher temperatureĬ) the two samples will reach the same temperatureĭ) the copper will reach a temperature lower than 25*CĮ) the silver will soften the silver will reach a higher temperature Which of the following quantities is not required to calculate the amount of heat energy required to heat water from 25*C to 55*C?Į) the heat of vaporization for water the heat of vaporization for water Raising the temperature of 10.0 g of water from 10.0*C to 20.0*C requires 100.0 cal of energy, while raising the temperature of 10.0 g of aluminum from 10.0*C to 20.0*C requires 22.0 cal. 100g of water by 10C How many calories are required to raise the temperature of a 35.0 g sample of iron from 25*C to 35*C? Iron has a specific heat of 0.108 cal/g *C.Į) 130 cal 38 cal The specific heat of copper is 0.0920 cal/g *C, and the specific heat of silver is 0.0562 cal/g *C. 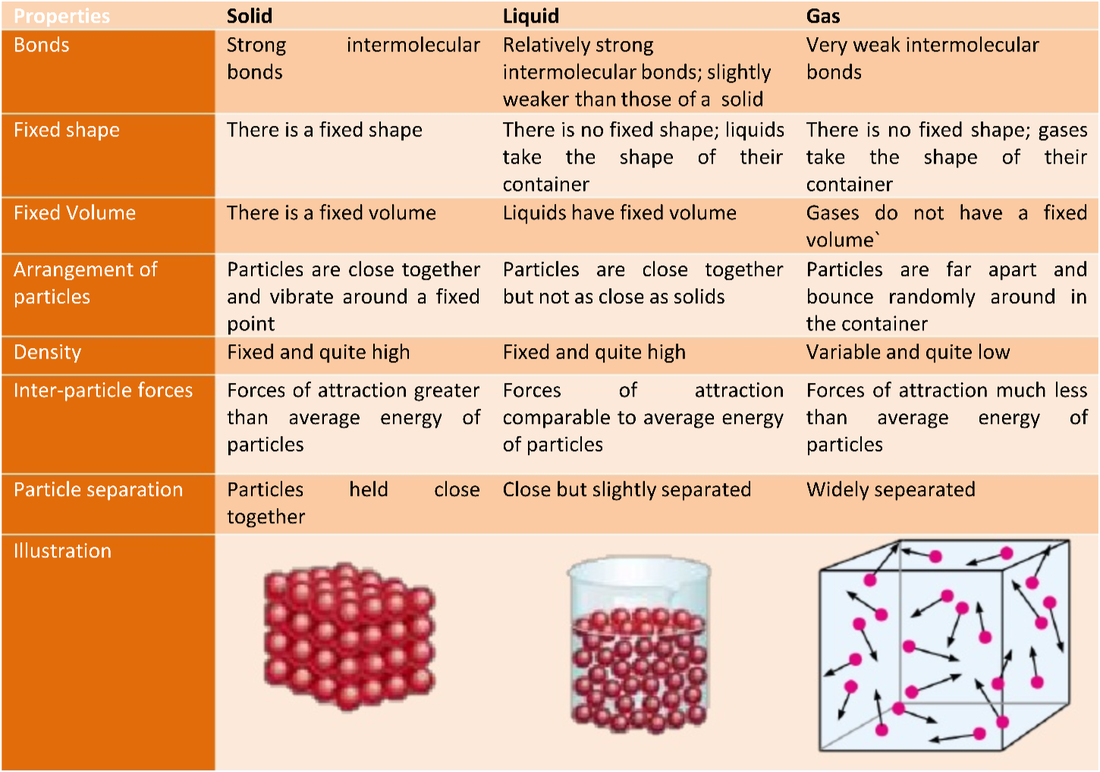
Round off the energy values for each food group to the tens place.)Į) 250 g 13 g The dietary calorie (Cal) is equal to _.Į) cal The specific heat of a substance is the amount of heat needed to _.Ī) change 1g of the substance from the solid to the liquid stateī) raise the temperature of 1g of the substance by 1*CĬ) change 1g of the substance from liquid to the solid stateĮ) convert 1g of a solid to a gas raise the temperature of 1g of the substance by 1C A kilocalorie of heat is required to raise the temperature of _.Į) 100 g of water by 100 ☌. If the pizza contains 280 kcal, how many grams of fat are present? (The caloric values are: 4 kcal/g for carbohydrate, 9 kcal/g for fat, and 4 kcal/g for protein. If protein has a caloric value of 4 kcal/g and fat has 9 kcal/g, how many kcal are in the serving? Round off the energy value for each food group to the tens place.Į) 490 kcal 240 kcal A slice of pizza contains 28 g of carbohydrate, 13 g of protein and fat. Round off the energy value for each food group to the tens place)Į) 520 kcal 240 kcal A serving of fish contains 50 g of protein and 4 g of fat. How many kilocalories, to two significant figures, does this sample contain? (The caloric values are: 4 kcal/g for carbohydrate, 9 kcal/g for fat, and 4 kcal/g for protein. If carbohydrate has a caloric value of 4 kcal/g, how many kcal are obtained from the carbohydrate in the potato?Į) 80 kcal 80 kcal One cup of kidney beans contains 15 g of protein, 1 g of fat, and 42 g of carbohydrate. J is the same amount of energy as _.Į) 2.72 cal 155 cal 3.25 kcal is the same amount of energy as _.Į) 13.6 J 13600 J A potato contains 20 g of carbohydrate. What is the temperature in degrees Celsius?Ī) the freezing point of water using the Celsius scaleĬ) the temperature on the Kelvin scale corresponding to 32FĮ) the freezing point of liquid nitrogen the coldest temperature possible An example of kinetic energy is _.Į) food running water The energy associated with the motion of particles in a substance is called _.Į) potential energy heat Which of the following is an example of potential energy?Į) riding an exercise bike water stored in a reservoir The phrase "ability to do work" is a definition of _.Į) cooling energy The energy stored in molecules is _.Į) calorie potential energy The energy of motion is referred to as _.Į) kinetic energy kinetic energy 650. What is the corresponding reading on the Kelvin scale?Į) 146 K 77K On a hot day, the thermometer read 95 ☏. What is the temperature in degrees Fahrenheit? (TF= 1.8(38.5)+32=)Į) 11.7 ☏ 101.3F The temperature of liquid nitrogen is -196 ☌. 
Which of the following is a property of both liquids and gases?Ĭ) has strong interactions between its particlesĮ) has large spaces between molecules has randomly arranged particles Which one of the following properties describes a liquid?ī) particles are close together and move randomlyĭ) fills the entire volume of the containerĮ) there is essentially no interaction between the particles particles are close together and move randomly In which of the following would the particles move most rapidly?Į) ice at 0C steam at 110C If the temperature is 20C, what is the corresponding temperature on the Fahrenheit scale? (TF= 1.8(20)+32=)Į) 94F 68F A patient has a temperature of 38.5 ☌.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
